| GENE SLOVERS US NAVY PAGES NAVAL ORDNANCE 1937 EDITION CHAPTER II SERVICE EXPLOSIVES |
| HOME INDEX History of Development of Explosives Explosive Substances-General Characteristics Explosive Mixtures and Explosive Compounds Uses of Military Explosives Propellants, Igniters, and Detonating Charges Manufacture of Smokeless Powder Reworked Powder Products of Combustion of Nitrocellulose Powder Stability of Nitrocellulose Powder Guncotton Black Powder Trinitrotoluene (TNT) Trinitroxylene (TNX) Picric Acid Tetryl Fulminate of Mercury |
| CHAPTER II. SERVICE EXPLOSIVES. Section I.-Brief History of the Development of Explosives. 201. The chemical constitution and physical form of modern explosives and the methods employed in handling and making use of them have resulted from a gradual development such as has been characteristic of the progress of so many other of the useful arts. A brief review of the more important phases of the history of the explosives art will be given as an aid to clearer understanding of the considerations which have governed the choice of the forms of explosive substances now used and their adaptation to present-day purposes. 202. Early incendiary substances.-Fire has, of course, been used as a weapon of war since the earliest recorded time and the transition from the use of ordinary combustibles to materials and mixtures of greater incendiary efficiency was a natural consequence of the gradual advance in physical and chemical knowledge. In the defense of Constantinople against the Moslems in the seventh and immediately succeeding centuries, the Byzantines made very effective use of the so-called “Greek fire,” particularly in naval engagements. This material was projected in flaming streams from tubes carried in the bows of the Byzantine vessels. It seems to have consisted of petroleum oils with the addition, perhaps, of sulphur. Similar materials were used both in streams of flame and in missiles by the Moslems during the Crusades, and knowledge of their use thus spread to other countries. Incendiary mixtures of this type were known during the Middle Ages as “sea fire” and “wild fire.” The early incendiary weapons were the forerunners of the modern flame thrower. 203. Black powder.-When the properties of saltpeter, or potassium nitrate, became widely known, about the thirteenth century, this substance was added to the earlier mixtures and a close approach was made to gunpowder. Charcoal soon came into use as the carbon-carrying constituent and the evolution of gunpowder was then complete. The use of saltpeter in incendiary mixtures seems to have been introduced by the Arabs and the Chinese at about the same time during the period referred to above. Knowledge of these mixtures, their uses and methods for their manufacture spread rapidly, especially after the first firearms were made, early in the fourteenth century. The gunpowder of these early times was, in its proportions as well as in its ingredients, much like the black powder in use today. Its history, through the centuries in which it remained the only widely known and used explosive, records development through changes of form and of methods of manufacture, rather than of chemical constitution. It was at first a very fine mealy powder. Later it was formed into rough grains, separated and graded into various sizes by sifting. Still later, compactness of the grains was secured by forming the powder into cakes under a considerable pressure before breaking it up into grains. For a long time gunpowder was used only as a propellant in small arms and in cannon. It then was turned to account in blowing up enemy fortifications and later for more peaceable forms of blasting. After powder-train fuzes had been successfully made, it came into use as a bursting charge for projectiles. In the early firearms, including cannon, the powder charge was ignited by the application of an open flame to a priming hole. The flintlock of the eighteenth century gave the first important improvement in methods of ignition. In this device the priming charge was ignited by sparks struck by the impact between flint and steel. Improved firing locks and the introduction of mercury fulminate came early in the nineteenth century and modern means of ignition have followed directly from them. Fulminate mixtures, in caps struck by a firing pin or in containers where they are ignited by the heating of electric bridges, are still the most widely used means of initiating the action which results in the burning of the charge of propellent explosive in a gun, or the detonating of a high-explosive charge in a projectile, a torpedo, or a mine. Although the compression and granulation of black powder had given a partial improvement in its performance in guns, it remained difficult to regulate the action, especially in larger ordnance. In attempting to increase muzzle velocities to get greater range and penetrating power, it was found that the gun chamber pressures soon became excessive because the powder burned too quickly. In 1860 General Rodman, of the United States Army, realizing the advantages to be gained by increasing the time of burning of the charge, proposed the use of large grains of very dense powder for this purpose. As a result of his researches he also proposed that perforated grains be used in order that the burning surface of each grain might be increased as combustion proceeded. The use of grains such as developed by General Rodman gave means of regulating much better the ballistic action of black powders, and such grains therefore came into general use. Various forms were common, such as the spherohexagonal and various prismatic shapes, including the hexagonal prism with a single perforation. The latter form was widely used in large guns. The use of such grains was the first notable advance in securing a powder which would burn progressively, that is, with increasing evolution of gases and heat. The last form of charcoal powder to be used in cannon was the “brown” or “cocoa” powder introduced about 1880. An underburned straw charcoal was used in this powder and gave it the characteristic color from which it took its name. This charcoal gave a denser and hence slower burning structure to the powder and permitted better regulation of pressure. 204. Cellulose nitrates and smokeless powders.-In 1838 Pelouze discovered that an explosive could be produced by nitrating cotton, that is, by treating cotton with nitric acid in such a way as to cause NO2 groups from the nitric acid, HNO3, to enter into combination with the cellulose of which the cotton is so largely composed. He thus produced cellulose nitrates, generally called nitrocellulose. His explosive was the first guncotton, but it was a very imperfect product and was not put to practical use. In 1845-46 Schonbein discovered that by nitrating cotton with a mixture of nitric and sulphuric acids an explosive of good quality resulted and that the nitration process could be controlled with some readiness. His process soon gained rather wide application since its importance in explosives manufacture was readily perceived. Manufacture of guncotton was undertaken in several European countries, but received severe setbacks through the occurrence of disastrous explosions in several factories in which it was being made. The researches of various investigators, notably of von Lenk in Austria and Abel in England, showed that the danger which had hitherto attended the manufacture of guncotton was due to the presence of impurities which could be removed by careful courses of treatment. The methods of purification which they introduced consisted principally in washing and boiling, together with pulping the material to facilitate cleansing. The earlier attempts to use guncotton as a propelling charge in guns were not successful. The velocity of the reaction was too great to permit of controlling the pressures, which were such as to burst many guns in which the explosive was used. Various measures were taken to retard the combustion, and cellulose nitrate powders, which were partially suitable for use in small arms, were produced by various makers. The solubility of cellulose nitrates in a mixture of ether and alcohol was noted by several who investigated its properties. The first to make successful application of this property of the material in producing a satisfactory propellent explosive was the French chemist Vieille, to whom was due a large part of the advance in knowledge of explosives. By thorough mixing of the nitrated cotton and the solvent he produced a gelatinous mass or colloid, which became quite hard and dense when the solvent was evaporated out. The resulting substance burned progressively and at moderate rates. The colloid was capable of being worked into the desired shape before drying and its formation into grains or strips to secure control of its burning was made possible. Vieille’s first powder of this kind, called Poudre B, was made in 1884. The improvements which have been made in smokeless powders since that time have been principally in the direction of better methods of purification and other measures for insuring chemical stability, although the technique of each of the other steps in the manufacturing process has also progressed in efficiency. 205. High explosives.-Nitroglycerin was discovered by Sobrero in 1846, but its highly explosive properties were not turned to accounts until Nobel, about 1860, found that it could be detonated by means of a small charge of fulminate of mercury. Nobel found also that nitroglycerin could be used effectively and with much greater safety when mixed with various absorbent materials. He began the manufacture of such mixtures, which have developed into many forms and have become widely known and used under the general name of dynamites. The nitration of cellulose and of glycerine to form powerful explosive substances led quickly to the discovery of a number of other explosives produced by the nitration of hydrocarbons, particularly of the aromatic hydrocarbons found in coal tars. One of the earliest additions to the list was picric acid, which had been known as a dyestuff long before its explosive properties were discovered and employed. Others have followed in great number and variety. Mixtures of various high explosive compounds with other substances, usually oxygen carriers, have also been developed and used, mostly for commercial purposes. The variety of widely known and used explosive substances has now become so large that the military explosives to be discussed in the remainder of this chapter comprise only a limited portion of the explosives field. Section II.-Explosive Substances: General Characteristics. 206. In the preceding chapter we have defined explosive substances and discussed the general characteristics of the reactions to which they give rise. In this section we will consider the characteristics of the explosive substances themselves and of the materials from which they are made. 207. Explosive mixtures and explosive compounds.-Regarded from the point of view of their composition, explosives may be divided into two classes: (1) explosive mixtures, (2) explosive compounds. Explosive mixtures consist of an intimate mixtures of distinct substances, properly prepared and conglomerated mechanically in varying proportions. Such explosive mixtures must have at least some oxygen supplier, such as a nitrate or chlorate, and some combustible, such as carbon or sulphur. Black and brown powders are typical examples of such mechanical mixtures. Explosive compounds consist of substances whose molecules contain within themselves the oxygen, carbon, and hydrogen necessary for combustion. They are true chemical compounds and are therefore homogeneous in constitution. They have weak molecular bonds, due to the presence in their molecules of weak bonding radicals, such as NO2. They are therefore in a state of unstable chemical equilibrium. Mechanical mixtures can be graded by varying the proportions of the ingredients. The elements constituting an explosive compound are always present in the molecule in the same quantities, according to the law of fixed proportions; therefore the nature of the explosive cannot be graded by varying the quantities of the constituent elements as in the case of mechanical mixtures. It is to be noted, however, that the same initial substance may, in many cases, yield different explosive compounds by nitrating to different degrees. The different products are, however, generally distinct chemical compounds. The explosive compounds consist very largely of nitrated hydrocarbons. The nitration results in the introduction of NO2 groups into the molecules of the hydrocarbon. The nitration is almost always effected by treating with nitric acid mixed with sulphuric acid. Most of the explosive compounds which are nitrated hydrocarbons are derived from hydrocarbons of the aromatic series. Most important of these basic hydrocarbons are benzene, toluene, zylene, naphthalene, anthracene, and their derivatives, all of which are found in the coal tars resulting from the distillation of coal to produce coke. Among the principal explosives derived from the aromatic hydrocarbons are trinitrotoluene and picric acid, together with the picrates. Of the explosive compounds derived by nitration of non-aromatic hydrocarbons, the most important are the cellulose nitrates and nitroglycerin. 208. Uses of military explosives.-Viewed from the standpoint of the military uses to which they are put, explosives may be divided into three classes: (1) Burning, progressive, or propellent explosives (low explosives). In this class are included all powders used to propel projectiles from guns. (2) Detonating or disruptive explosives (high explosives). This class includes the explosives used f or bursting projectiles and for the main charge in torpedoes, mines, aerial bombs, and for most demolition purposes. (3) Detonators or exploders. There are high explosives used in small quantity to initiate explosive reactions in charges of explosives belonging to the two classes above. 209. Propellants.-Smokeless powders of one form or another are now used almost universally for propellent charges. For military purposes, especially for guns larger than small arms, they may be considered to be of two classes, (a) single base powders and (b) double base powders. In the single base powders, cellulose nitrates, which will hereafter be referred to as nitrocellulose, form the only explosive ingredient. The other materials present in single base powders are present to give suitable form and stability to the powder. In the double base powders, nitroglycerin is present to assist in dissolving the nitrocellulose during manufacture, as well as to add to the explosive qualities of the powder. The single base nitrocellulose powders contain proportionately less oxygen than the double base powders. The resultant gases from the single base powders contain therefore relatively less of carbon dioxide, CO2, and relatively more of carbon monoxide, CO, than do the double base powders. The heat liberated by equal weights of the two powders is therefore greater in the case of the double base powders, since the heat of formation of CO2 is greater than the heat of formation of CO. The conversion of carbon to carbon monoxide produces a greater volume of gas than its conversion to carbon dioxide. The single base powder therefore produces a greater volume of gas, though less heat than the double base powder. From a thermodynamic standpoint, it is therefore somewhat less efficient, but it has the advantage of causing less erosion in the gun because the resulting temperatures are lower. Single base powders are used in France and in the United States. Double base powders containing nitrocellulose and nitroglycerin are used in England as “Cordite,” in Italy, in Spain, and various other countries. 210. Igniters.-Charcoal powders are now no longer used as propellants in military rifles. In the form of black powder, they are now used to facilitate ignition of smokeless powder charges, since they themselves are more readily ignited; in fuzes, to propagate flame from one part to another; for saluting charges; for bursting charges in certain classes of projectiles; and for various other purposes. 211. Detonating charges.-Large charges of high explosives are used as the main charges of torpedoes, mines, aerial bombs, and various classes of projectiles. The explosive most commonly used for these purposes during recent years has been trinitrotoluene, generally known as TNT. This material has also been called trinitrotoluol, trotyl, trinol, trilite, tritolo, and by various other names. For a number of years prior to the World War, guncotton was the preferred explosive material for torpedoes and mines, whereas picric acid or its derivatives were used commonly for the bursting charge of projectiles. TNT has supplanted guncotton because of its greater safety, stability, and convenience. It has also replaced picric acid and the picrates for filling certain classes of projectiles, especially those not intended for piercing armor plate. TNT was used in such quantities during the war that shortages in the materials required for its manufacture were threatened. This led to efforts to find satisfactory materials which could be mixed with TNT in order to conserve the available supply and yet secure powerful explosive mixtures. These efforts led to the adoption in various services of such explosives as amatol and sodatol, which consist of mixtures of TNT with ammonium nitrate and sodium nitrate, respectively. The material adopted by the United States Navy for mixing with TNT was trinitroxylol, or TNX, which is closely related to TNT. Xylene and toluene, the substances from which these explosives are obtained by nitration, are, as previously mentioned, both derived from coal tar. Picric acid and the picrates are still largely used as bursting charges for projectiles since they possess advantages over TNT for certain classes of projectiles. They are preferred for armor-piercing projectiles because of the fact that they do not deflagrate in passing through armor plate as readily as does TNT or give incomplete explosions. Since an armor-piercing projectile will be more effective when bursting inside an armored structure after penetration than when bursting in passing through the armor, the characteristic discussed above becomes of much importance in selecting bursting charges. Picric acid and its derivatives have been used in various countries under the name of lyddite (English), melinite (French), explosive D (United States), shimose (Japanese), and ecrasite (Austrian). TNT has been used extensively as the bursting charge in high-explosive projectiles not intended to be fired against armor. The general requirements of high explosives for projectiles, applying particularly to armor-piercing projectiles, are given below: (1) Should be reasonably safe to manufacture, and free from injurious effects to the operators as far as possible. (2) Must show a safe degree of insensitiveness in ordinary handling. (3) Must withstand the maximum shock of discharge from the gun in the projectile for which it is intended. (4) Must withstand the shock of impact without explosion when fired in fuzed projectiles against the strongest plate that the projectile alone will perforate without breaking up. (5) Must be uniformly and completely detonated with the service detonating fuze. (6) Should possess the greatest explosive power compatible with other necessary requirements. (7) Must not decompose when a dry or wet sample is hermetically sealed and subjected to a temperature of 65.5° C. for one week. (8) Should be non-hygroscopic, and must not have its facility for detonating affected by moisture that can be absorbed under ordinary atmospheric conditions of storage and handling. High-explosive charges are usually loaded by melting and pouring if the kind of explosive substance used permits of this treatment. This gives greater density to the charge and hence greater explosive effect in a container of given volume. TNT lends itself especially to the casting of charges. 212. Detonators.-A discussion of the function of detonators, including boosters, was given in the preceding chapter. Detonators are of a great variety, in both mechanical and explosive details. The detonating materials used in detonators and boosters are chosen for their efficiency in detonating the explosive substances of which the main charge is composed. The detonating reaction itself begins with a small charge of fulminate or fulminate mixture and is transmitted through the booster to the main charge. The substances now most commonly used for the detonating trains and for boosters are tetryl, picric acid, and crystalline TNT. Section 111.-Manufacture and Characteristics of Service Explosives. 213. In this section will be given brief notes on the manufacture of certain explosives now used in the United States Navy, with further reference to the properties and characteristics of each. Inasmuch as the details of manufacturing methods and equipment change somewhat from time to time and vary to some extent between different manufacturers, such details will in general not be given fully. Attention will be directed more closely to the principles followed in manufacture and the precautions taken to insure safety in manufacture and purity and stability of the product. SMOKELESS POWDER 214. The smokeless powder used by the United States Navy is a uniform ether-alcohol colloid of carefully purified nitrocellulose. A small quantity of diphenylamine is added in the course of manufacture to assist in preserving the chemical stability of the powder. The powder is made to conform to very rigid manufacturing specifications, which insure a very pure product, and to rigid ballistic specifications, which insure uniformity of performance in the gun. Except for minor differences in specifications, the same kind of powder is used by the United States Army. 215. Raw materials.-The principal raw materials used in the manufacture of United States Navy smokeless powder are the following: (a) Cotton.-The cellulose material to be nitrated consists of bleached and purified unspun cotton wastes or short-fibered cotton, the latter being obtained through the removal of the fiber ends which are found adhering to the cotton seed after the ginning process. Shortfibered cotton is particularly suitable for nitration because the nitrating acids can reach all parts of it readily, which tends to uniformity in nitration. The purification to which the cotton has been subjected before it is received at the powder factory consists usually in boiling with caustic soda to remove impurities, especially the waxy constituents of the fiber, and to make those which remain more readily removable in the purifying processes undertaken later during the course of manufacture. The cotton is then bleached with chlorine, washed, and dried. (b) Acids.-A mixture of nitric and sulphuric acids is used in the nitrating process. Mixed acids as made or received at the factory are required to conform to rigid specifications as to purity and strength. The mixture as received has a total acid content of about 95 per cent, and the proportion of nitric and sulphuric acids is about equal. The acid as used directly in the nitrations is mixed with weaker acids which have been used in previous nitrations, in which they have lost a part of their acidity. These weaker recovered acids are known as spent acids and the process of mixing them with the fresh acid mixture is known as fortifying the spent acids. The usual acidity of the nitrating mixture is about 85 per cent, but this is varied somewhat with changing conditions, especially with changes in temperature. The proportion of the acid constituents is about two parts of nitric acid to one of sulphuric acid, by weight. (c) Ether and Alcohol.-A mixture of ethyl ether and ethyl alcohol is used as a solvent for the nitrocellulose. They are required to be of a high purity. (d) Carbonate of soda.-Carbonate of soda is used in the water in which the nitrocellulose is boiled during certain stages of the purification process. It also is required to be of a high degree of purity. (e) Diphenylamine.-Diphenylamine, a pale yellow crystalline organic substance with a slightly alkaline reaction, is incorporated with the powder in order to neutralize any acid products which might be formed in the powder as a result of gradual decomposition. Diphenyla mine thus prevents decomposition from becoming progressive and hence adds to the chemical stability of the powder. It is therefore called a stabilizer. Other substances with similar chemical characteristics and reaction are available as stabilizers and have been used in various countries. |
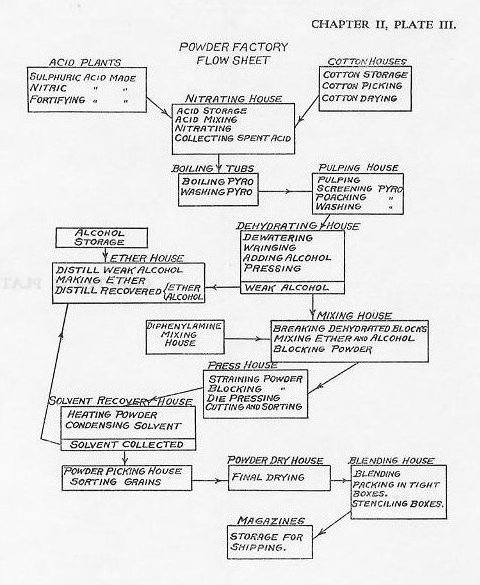
| 216. The principal steps in the manufacture of smokeless powder (Plate III) are as follows: (a) Picking of cotton.-The purified cotton is passed through a picking machine which tears apart the knots and tangled lumps. (b) Drying of cotton.-The cotton is next passed through driers which reduce the moisture content to about one per cent. Moisture in the cotton during the nitration process would tend to non-uniformity of nitration and would increase the danger of burning the material due to the heat involved by the interaction of the acids and the moisture. After drying the cotton is weighed in cans and, when cooled, carried to the nitrating house. (c) Mixing acids.-The mixture of sulphuric and nitric acids used in nitration is obtained by fortifying spent acids. The mixture is brought to a temperature of about 30 degrees C. in heating tanks before delivery to the nitrators. (d) Nitrating.-Approximately 60 pounds of cotton are put into each nitrating pot containing the mixed acids. The nitration process takes about 20 minutes, during which the cotton and acids are thoroughly agitated by revolving paddles. The cotton is converted into nitrocellulose containing approximately 12.60 per cent nitrogen. This is commonly called “pyrocellulose.” Each charge of nitrocellulose and excess acid is then run into a wringer below the dipping pot, where the spent acids are removed by centrifugal force. During nitration it is generally accepted that the first chemical action is a conversion of the cotton into cellulose sulphates, with the liberation of water, by the action of the sulphuric acid. The nitric acid then, acting upon the cellulose sulphates and displacing the sulphuric acid constituents, produces cellulose nitrates. The liberated sulphuric acid combines with the water resulting from the first reaction to form hydrates. The replacement of the cellulose suiphates by cellulose nitrates, however, is not complete and a certain proportion remain as impurities to be removed in the subsequent purification processes. (e) Purifying.-(1) Drowning.-The pyro is forked from the wringers as quickly as possible to drowning troughs of running water. If it should be exposed to the air for any considerable period, it is likely to take fire through the action of the nitrating acids not completely removed in the wringing process, and to burn slowly with an evolution of dense nitric fumes. The pyro, completely immersed, is run by pumps or by gravity to the boiling tubs. During this transfer most of the free acids remaining in the pyro pass off into the water. (2) Preliminary boiling. The pyro is boiled for about 40 hours in the boiling tubs, the water being changed about five times, to further remove the free acids. After this it is transferred in a trough of running water to wooden boxes with false bottoms to permit drainage, which are located over the pulpers in the pulping and poaching house. (3) Pulping. The pyro is cut and ground in the pulpers to permit the next stage in purification to reach all parts of the fiber, and to reduce it to a consistency desired in later operations. (4) Poaching. The pyro is next boiled in water in poaching tubs with an addition of carbonate of soda, and then several additional times in water alone, the water being replaced between boilings. The poaching tubs are large tubs, similar to the boiling tubs, each holding about 3600 pounds of equivalent dry powder. After this final course of purification a sample of the poacher lot is chemically tested. If sufficiently pure, the lot is pumped to stock tubs in the dehydrating house. (f) Dehydrating.-The pyro is transferred to the dewaterer and to centrifugal wringers which remove the excess water, after which a charge of about fifty pounds is loaded into the cylinder of an hydraulic press, where the remaining water is forced out by forcing alcohol into the cake under pressure. The pyro cake, so formed, is subjected to a final pressure to remove the excess alcohol, leaving only sufficient alcohol for making the desired colloid. (g) Mixing-The compressed cake, in a metal can, is taken to the mixing house, where it is first broken up in a rotating drum or block breaker into a fine material which lends itself more readily to the mixing operation and hence insures a more uniform colloid. The pyro is then transferred to the mixer where the ether constituent of the solvent and the diphenylamine stabilizer are added. The charge is mixed for about half an hour, during which it becomes partially dissolved or colloided by the ether and alcohol. It is then reformed into a block in an hydraulic press and carried to the press house. (h) Pressing.-The pyro block is forced through small holes of a strainer (macaroni) press to more thoroughly mix and to give greater homogeneity to the colloid. It is again reblocked and then forced through the die press, from which it emerges in the form of a continuous cord of the desired diameter with seven perforations (one in small grain powder). The cord is led to a machine cutter which cuts it into grains of the desired uniform length. The "green" powder, as it comes from the die press, is fairly soft because of the large excess of solvents which it contains. It is carried in boxes to the solvent recovery and dry houses. (i) Recovery of solvents.-The powder is subjected to a heat treatment for several days, the length of time depending upon the size of the powder, to evaporate a portion of the excess solvents, which are condensed for separation and further use. After the solvents are evaporated, the powder grains become hard and translucent, and very much like horn. The powder is next conveyed to the powder picking house. (j) Picking.-The powder is picked over by hand to remove all grains of abnormal size or appearance. (k) Drying.-The green powder is next dried in large dry houses at air temperature for about 60 days and then in a constant circulation of air at about 40° C. from 2 to 4 months. The duration of drying will depend upon the total amount of volatiles to be evaporated, and upon the size of the grain. When the total amount of volatiles is reduced to the proper percentage, the grains will have contracted to the desired dimensions to give the proper speed of burning. The powder shrinks materially during drying. The final dimensions of the grains exert an important influence upon the ballistic characteristics of the powder in a given gun. The percentage of volatiles remaining in each powder after drying varies from three per cent to seven per cent, being greater in the larger granulations. Nitrocellulose smokeless powders also can be dried by circulating through them warm water instead of warm air. The temperatures in water drying are somewhat higher than in air drying, and the process is quicker. (1) Blending.-A number of poacher lots, each of which maintains its identity until passed through the dry houses, are blended to make up one uniform lot of powder. After the drying is completed and before blending, the powder is exposed to the atmosphere for from 24 to 60 hours in order to insure that surface moisture is as nearly uniform as possible. Blending is usually carried out in blending towers. The tower consists of a series of bins arranged in groups, with groups one above the other. The bins of each group are usually arranged in polygons and each bin has a trap or gate valve in the center of its group. In blending, the top group of bins is filled with powders from different poacher lots. The traps or gate valves are opened simultaneously and the powder from the bins falls to the next group in one stream, which separates about equally into the bins of the next lower group. This operation is continued from group to group and the powder is finally drawn off through a hopper at the bottom of the blending tower into the powder boxes. A sample is taken for complete chemical tests. The powder boxes are stenciled with the powder designation, the place of manufacture and the serial number of the lot. For example: IHHA Lot 40 means powder manufactured at Indian Head (IH) for 14“/50 (HA) cal. gun and that it is lot 40 of this particular caliber. Various letters such as D for DuPont, I for International, etc., designate the different manufacturers. The last one or two letters indicate the caliber and are published in a table in the powder specifications, for example, H stands for a 14”/45, H.A. for a 14"/50; C stands for 6”/40, C. B. for 6”/50, C.C. for 6”/53. (m) Proof and disposition.-A firing sample for proof at Dahlgren is selected from the lot of powder and the balance is stored in a magazine until ordered shipped by the Bureau of Ordnance. On acceptance, the Bureau gives the lot of powder a service index number which is assigned in regular numerical sequence in the order of acceptance. These indexes are also designated by certain letters, for example SPDW 2132. The SP indicates smokeless powder, the D stands for diphenylamine (showing it is a stabilized powder), the W stands for reworked, the number is the index number. Other letters used are R for rosaniline (used in 1908-09 as a colored indicator of the stability of the powder but since abandoned), B for a blend of two or more indexes, X for water dried (instead of the usual air drying), and F for flashless powder. 217. Manufacture of reworked smokeless powder.-Unstable powder from the service, odds and ends of powder from magazines, powder accidentally wet, in fact, any unserviceable powder is sent to Indian Head for reworking. On receipt, it is dumped into concrete vats and kept under water. In reworking, the first steps are grinding and purifying. The old powder is ground under water to a fine pulp, similar to the fresh pulp coming from the pulpers, by a pair of heavy, iron runners or wheels traveling around a circular iron bed. The rollers are seven feet in diameter, weigh about nine tons each, and have a common horizontal shaft about which they turn. From this point on it is put through the same general processes as new pyro. However, if only diphenylamine powder is ground, the boiling treatment is generally omitted. In any event the material must pass the usual tests for pyro before it is sent to the dehydrating house. Throughout the manufacture, the lot number carries the letter R for reground pyro in order to distinguish it from new pyro lots. When blended into a powder lot and after proof, the index is assigned a designating letter W to indicate that it is reworked powder. Reworked powders are considered nearly as good as new powders, the principal difference being that their nitration is not quite so high, and the grains are not quite so tough. Reworked powders are darker in color than new powders. 218. Products of combustion of nitrocellulose powder.-The nitrocellulose constituent of the powder is regarded as a mixture of enneanitrocellulose, C24H30O20(NO2)9, and deca-nitrocellulose, C24H30O20(NO2)10. These nitrocelluloses are the highest which are soluble in the ether-alcohol mixture. The powder burns regularly and progressively, leaving an almost negligible quantity of ash. The decomposition of our nitrocellulose powders in burning is usually represented by the following equation: C24H30(NO2)10O20 = 12C02+ 12C0 +4H20 + 11H2 + 5N2. This represents a reaction which is complete for a nitrocellulose of 12.75 per cent nitration. It does not take into account the reactions due to the presence of the volatiles or the stabilizer. It is deduced from theoretical considerations and from the study of products of explosion made in laboratory apparatus in which only a small amount of explosive is used, the density of loading being therefore small, and in which the gases have been considerably cooled before examination. That the actual products of combustion differ considerably from those above represented is evidenced by the deep orange-colored cloud of gas accompanying the discharge of the gun. This color is due to oxides of nitrogen, which are not accounted for in the theoretical equation. Since the actual products of combustion are not all colorless gases the powder cannot be called truly “smokeless.” This term has been used in a relative sense to distinguish the modern powders from their predecessors which gave off dense clouds of white smoke. The gases from “smokeless” powders are much less opaque and are dissipated much more rapidly. A small cloud of white smoke, due to the black powder ignition charge, is usually readily distinguished in the gas from a gun firing smokeless powder. It will be noted that carbon monoxide forms a considerable portion of the products of combustion in the above reaction. The effect of this upon the temperature of the gases during the explosive reaction has already been pointed out, as has also the possibility of “flareback” due to ignition of this inflammable gas when mixed with air. 219. Stability of nitrocellulose powders.-Powders containing nitrocellulose are subject to a very gradual chemical decomposition which may in time be a source of danger unless measures are taken to arrest or check such action. From its nature nitrocellulose is, like many explosive compounds, in a state of unstable chemical equilibrium and is readily acted upon unfavorably by impurities which may be present with it. If decomposition takes place in any particle the decomposition products will include nitrogen oxides which have an acid reaction and will facilitate further decomposition. The reaction occurring will be accelerated for this reason and a progressive decomposition will result. The decomposition will be greatly facilitated by heat and by the presence of moisture. During manufacture every precaution is taken to insure the absence of impurities in the raw materials and the removal of impurities during the various courses of purification. The principal impurities removed during manufacture are the suiphates, which are formed as by-products, so to speak, of the nitrating reaction, and the free acids remaining after nitration. The addition of the stabilizing substance, diphenylamine, is the means taken to counteract any decomposition which may begin in the finished powder. The way in which diphenylamine arrests decomposition through neutralizing the acid decomposition products has already been mentioned. Progressive decomposition cannot occur until all of the diphenylamine in the powder has been used up by combining with the acids. The use of diphenylamine has greatly increased the stability life of our powders and the limits of its effectiveness have not yet been reached. The presence of residual volatiles in the powder also tends to retard possible decomposition. For this reason, as well as to avoid change in ballistic qualities through change in the rate of combustion, every effort is made to prevent the loss of volatiles from the finished powder. Excessive heat will have a most unfavorable influence upon the stability of the powder. At temperatures below 60° F. the stability is not appreciably affected. At temperatures above 70° F. the rate of decomposition rises quickly with rises in temperature, becoming high at 90° F. and dangerously accelerated at temperatures over 100° F. Precautions are therefore taken to insure the maintenance of a uniformly low temperature as far as possible in the magazines in which powder is kept on shipboard and elsewhere. Since the presence of moisture favors decomposition, the containers in which the powder is stowed are made air-tight and every effort is taken, through care in handling, to maintain their tightness. A leaky container may not only admit undesirable moist air to the powder, but may permit the loss of volatiles through evaporation, especially if the air in the container is changed through alternate expansion and contraction due to changes in temperature. 220. Loss of volatiles.-Loss of volatiles, through leaky containers or otherwise, will increase the rate of combustion of the powder; hence it will increase the pressure in the gun due to a given weight of charge. It is possible that a charge which has lost volatiles to a considerable extent may produce dangerously high pressures when fired. Such powder has sometimes been said to be dynamically dangerous. “Ballistically dangerous” would probably be a more descriptive term. It is readily seen that the same precautions-tight containers and uniformly low temperatures-which guard against the development of “unstable or chemically dangerous” powders will also guard against the development of ballistically dangerous powders. Under normal conditions of storage powders will not lose volatiles to any appreciable degree. A powder which may have become dangerous chemically through partial decomposition is not dangerous ballistically since a part of the decomposition, which should take place in the gun with sudden evolution of heated gases, has already taken place and the powder has lost a corresponding number of heat units. GUNCOTTON 221. (a) Properties-Guncotton is composed of nitrocelluloses of higher degrees of nitration than those which are used for smokeless powder. In appearance it is very much like ordinary white cotton when pure. When wet it retains its chemical stability for long periods during storage. It is quite insensitive to flame, shock, or friction. It can, however, be detonated, even when wet, by means of a booster of TNT or dry guncotton. Dry guncotton is much more sensitive and less stable chemically than wet guncotton. (b) Manufacture.-The manufacture of guncotton in general is similar to the manufacture of nitrocellulose for smokeless powder, except that the nitrating process is more sustained in order to produce a higher percentage of nitration. After purification of the nitrocellulose the guncotton is usually pressed into blocks of the desired form. Wet guncotton contains water in the amount of about 25 per cent of the dry weight. (c) Uses.-Wet guncotton was for many years the principal explosive used in torpedoes, mines, and demolition charges. Dry guncotton was used as a booster for wet gun cotton charges, and is still used as a flame producer in certain types of primers. Due to its sensitivity when dry, its liability to deterioration, its low power as compared with other high explosives, and the availability of raw materials for the aromatic compounds, namely, derivatives of coal tar, guncotton has been superseded as a naval explosive. BLACK POWDER. 222. (a) Properties.-The composition of black powder varies somewhat, depending upon the use for which it is intended. The usual composition, however, is about as follows: Saltpeter (potassium nitrate), 75 per cent; charcoal, 15 per cent; sulphur, 10 per cent. The form of the powder also differs, depending upon the use for which it is intended. When made up in grains, the powder has a somewhat glazed surface and is free from dust. For certain special purposes, for instance in fuzes, a very fine black powder known as fuze powder or meal powder is used. Black powder possesses practically unlimited stability if stored in tight containers and kept perfectly dry. It deteriorates irregularly when exposed to moisture; consequently, all ammunition containing it, such as powder-train time fuzes, must be protected from a moist atmosphere. It is not affected by moderately high temperatures, nor is it subject to spontaneous combustion at ordinary storage temperatures. It is highly inflammable and is sensitive to friction, shock, and sparks of heat. It is extremely quick and violent in its action when ignited. A container of black powder should never be opened in a magazine or room having other explosives, except when assembling ammunition in which other explosives are sealed. The maximum limit of exposed black powder, except in manufacturing operations, is placed at 25 pounds. (b) Manufacture.-The ingredients are powdered by grinding separately before mixing. A preliminary mixing is then given, usually in a rotating wooden drum containing lignum vitae balls. The mixture is usually thoroughly incorporated in a mill in which it is ground and worked under large slow-moving rollers. In this process the mixture cakes somewhat and it is therefore broken by hand before passing to the press. In the press the powder mixture is subjected to high pressure to produce a dense, hard mass. The pressed mass is broken up and passed through a granulating mill where it is broken into smaller grains by passing through several pairs of rolls. These grains are separated into different sizes by passing through sieves. The grains are glazed by being tumbled about in rotating wooden drums. Glazing stops up the pores of the grain, thus making the powder less sensitive to moisture and less apt to deposit dust. Some powders, especially larger grain sizes, have graphite added to facilitate glazing. After glazing the powder is usually dried under moderate temperatures. The various grades of black powder are classified according to their percentage of composition, purity of material, size of granulation, and rate of burning. (c) Uses.-The following granulations of black powder, arranged in order of decreasing size of grain, are now in use in the naval service: (1) Spherohexagonal for use as torpedo impulse charges. (2) Cannon powder for use in ignition pads of smokeless powder charges for bag guns and for saluting charges. (3) Shell powder for use as burster charges of projectiles, either alone or in combination with a high explosive as mixed filler. (4) Fine grain, designated as FFG and FFFG, for use in primers and illuminating projectiles. Other black powders are used in Very signals, rockets, other pyrotechnics, base charges for shrapnel, time train rings of fuzes, pellets for delayed action fuzes, etc. TRINITROTOLUENE (TNT) 223. (a) Properties.-Trinitrotoluene, frequently called trinitrotoluol, or TNT, is a crystalline solid. ‘When fairly pure it varies from white to pale yellow in color and is known as grade A, or refined TNT. When the proportion of impurities is greater the color is darker, being often brown, and it is known as grade B, or crude TNT. At the present time the following grades and forms are found in the Navy: Recrystallized (purified crystals), flaked grade A, granular grade A, and granular grade B. Any of these grades may be melted and cast to become cast TNT, grade A; or cast TNT, grade B. The differences between grades is one of purity and of melting point. The specific gravity is about 1.64. TNT is nearly insoluble in water. It is practically non-hygroscopic and absorbs moisture only to a negligible degree when exposed to damp atmosphere. A cast charge of TNT is impervious to water if its surface is unbroken. TNT in any form may remain under water or exposed to water for long periods without harmful effect, and, when dried out, should be as efficient as before. Water present with granular TNT will, however, affect the readiness and completeness with which it is detonated. When the amount of water reaches about 15 per cent by weight the granular TNT cannot be detonated with the detonators usually used. TNT is neutral in its reaction and does not form sensitive compounds by reaction upon metals, as does picric acid. It is chemically stable and will retain its stability for long periods even under variable and unfavorable conditions of storage. In fact its stability life seems almost unlimited. It is relatively insensitive, although it may be ignited by impact, friction, spark, shock, or heat. It is readily melted, the melting point varying between about 80.6° C. for grade A and 76° C. for grade B, and cast into bursting charges for projectiles, bombs, etc. In a granular or crystalline form, and when unconfined, it burns freely and may burn completely without detonation. In the cast form it will almost invariably detonate in a fire. Grade A TNT is detonated more readily than grade B, and granular or pelleted TNT is much easier to detonate than cast TNT. A granular or pelleted booster charge of grade A TNT or of tetryl is therefore usually used to detonate charges of cast TNT. Thin-walled projectiles, bombs, depth charges, mines and warheads, containing TNT bursting charges, are subject to sympathetic detonation. This property necessitates TNT storage separate from other ammunition. The disruptive force of TNT has been stated as roughly 10 per cent less than that of picric acid, and slightly greater than that of wet guncotton. When detonation is complete it gives off a considerable volume of black smoke, due to the uncombined carbon resulting from the reaction. When detonation is incomplete the smoke given off is yellow or yellow gray due to the presence of unconsumed TNT. The rate of detonation of TNT is about 7,000 meters per second. Unless very pure, TNT usually contains a certain proportion of isomers (that is, substances having the same chemical formula but with a different molecular arrangement). These isomers have a lower melting point than the pure TNT and they therefore tend to segregate and exude, especially from “crude” TNT, and more particularly from cast charges. This exudation increases with increased temperature. The appearance of these isomers, exuding from TNT containers as dark brown oily liquids, is not uncommon and need not cause alarm. The isomers are detonated less readily than pure TNT and their presence, therefore, affects the readiness with which the whole charge is detonated, but it appears that they take part in the reaction when the whole charge detonates. The isomers are inflammable to about the same extent as TNT and are not more sensitive to impact or friction. Their presence does not appear to affect chemical stability. (b) Manufacture.-Trinitrotoluene is produced by the nitration of toluene. This substance is one of the oily liquids present in coal tar and separated from it by fractional distillation. The nitration is carried out by treating toluene with a mixture of nitric and sulphuric acids at a temperature of about 160° F. The reaction proceeds in steps, the initial product being mononitrotoluene, which by further nitration is converted to dinitrotoluene, and finally to trinitrotoluene. The progressive nitration is sometimes carried out in a single container or kettle, the successive steps being brought about by the addition of fresh acids to strengthen or fortify the mixture; it is sometimes carried out by conducting the three steps in three sets of nitrators, the third step requiring the strongest acids. After the spent acid from the last step in the nitration has been drained off the crude trinitrotoluene is crystallized by melting in strong sulphuric acid and cooling. The acid carries off impurities in solution and the trinitrotoluene crystallizes out in cooling. The crude trinitrotoluene is washed several times with hot water until the remaining acids are removed. It is then melted in a steam jacketed pan and heated until the water is driven off, and recrystallized by cooling while being stirred constantly. Purification and recrystallization is often carried out by treating with various solvents other than sulphuric acid, such as alcohol, alcohol and carbon tetrachioride, and sodium sulphite solution. The methods used differ with various manufacturers. During the manufacture of trinitrotoluene and its subsequent handling workers are often subject to a slow poisoning through inhalation of vapors and dust, or absorption of material through the skin. The effect of this poisoning usually wears off when exposure is stopped and can be prevented or much reduced by the wearing of gloves and protective clothing, and by washing the clothing and body carefully. (c) Uses.-(1) A refined granular or crystalline TNT of high melting point is used for booster charges, fuzes, and preferably for burster charges of projectiles. However any grade may be used for the latter. TNT is not sufficiently insensitive to be used as burster charges for armor piercing projectiles. (2) Cast TNT of one of the lower grades is ordinarily used for the main charges in mines, warheads, bombs, and depth charges. (3) TNT is frequently used as a component in other explosives. When mixed with black powder as a projectile burster charge, it increases the bursting effect of the black powder. TRINITROXYLENE (TNX) 224. Trinitroxylene or TNX is produced by the nitration of xylene, which like toluene is obtained from coal tar. This substance is a brown crystalline solid, whose physical and chemical characteristics are analogous to those of TNT, and whose stability and safety are comparable to those of the latter. It is detonated with more difficulty than TNT, but in mixture with the latter in proper proportions gives an explosive substance which is practically as satisfactory for cast charges. (See Art. 211.) PICRIC ACID 225. Trinitrophenol or picric acid is derived from phenol, otherwise known as carbolic acid. It is a yellow crystalline solid of good chemical stability and highly explosive properties when properly detonated. It has good chemical stability except that it is prone to act upon metals to form metallic picrates which are very sensitive explosive substances. For this reason, except as a raw material in the manufacture of ammonium picrate, explosive D, it is not used by the U. S. Navy. (a) Manufacture.-Picric acid manufacture is usually carried out in two stages. In the first stage phenol is treated with sulphuric acid. This suiphonation results in the production of phenolsuiphonic acid, which is nitrated with nitric acid to form trinitrophenol. The product is carefully purified by washing with water. (b) Uses.-Picric acid has been used as a booster material in various types of fuzes, and under the name of melanite and lyddite has been extensively manufactured by the English and French governments, respectively, for use as a bursting charge for projectiles. It has also been mixed with TNT to form an explosive for use in mines. AMMONIUM PICRATE (EXPLOSIVE D) 226. (a) Properties.-This explosive, which was patented in 1888, was for many years the secret high explosive of the United States. As such it was known as explosive D. Its particular importance as a military explosive is due entirely to its marked insensitiveness to shock and friction. It is only slightly inferior to TNT in explosive strength. It is a crystalline powder of light yellow color which is loaded in projectiles by tamping. It does not melt when heated. It is only slightly hygroscopic, but when wet forms sensitive and dangerous picrates with copper and lead. It does not form dangerous compounds with steel, but, to avoid corrosion, the interiors of projectiles are covered with varnish before being filled. It has high chemical stability even when subjected to temperatures as high as 150° F. for considerable periods of time. When ignited and not confined, it burns slowly, with dense black smoke. If confined and heated to the ignition temperature, it will explode. It can hardly be brought to detonation by blows and requires a strong detonation to explode it. (b) Manufacture.-Ammonium picrate is made by saturating a hot solution of picric acid with aqua ammonia or ammonia gas. This results in neutralizing the acid, which is shown by the formation of crystals. The solution, when the reaction is complete, is dumped into crystallization tanks, where the ammonium picrate crystallizes out. The crystals are then removed, drained, dried, and screened. The powder is then ready for packing. (c) Uses.-Explosive D is used primarily as the burster charge for large caliber projectiles, especially armor piercing projectiles, as it will withstand the shock of impact against any thickness of armor if properly loaded. The advantage of this is, of course, that the armor piercing shell will have partially or completely entered the plate before the shell filler will detonate as a result of the fuze action. It sometimes is used to form a mixed filler with black powder for projectiles with ignition fuzes. TETRYL 227. (a) Properties.-Tetryl, or tri-nitro-phenyl-methyl-nitramine, a yellow crystalline explosive substance, sometimes called tetra-nitro methyl-aniline, is usually produced by the suiphonation and nitration of dimethylaniline, which is produced from aniline and methyl alcohol. The product is purified by washing in water, drying, and recrystallizing from hot benzol. Tetryl is more powerful, as well as more sensitive, than either picric acid or TNT. It is poisonous in character. It is stable at all ordinary temperatures. Its strength and brisance would seem to adapt it for use as a bursting charge, but its sensitiveness is such that if used as a shell filler, it would not stand the shock of discharge. (b) Uses-Tetryl is a standard booster charge explosive. It is also used in the detonating trains of fuzes and as a constituent of detonators. For detonators and blasting caps it is mixed with fulminate of mercury; the latter mixture is more powerful than a mixture of TNT and fulminate or picric acid with that of fulminate. FULMINATE OF MERCURY 228. (a) Properties.-Fulminate of mercury is a fine yellowish-white crystalline powder. The gray color of the commercial fulminate is due to small particles of unconverted mercury. When dry it is very sensitive and is readily detonated by percussion, friction, spark or flame, or by contact with sulphuric or nitric acid. When moistened its sensitivity is much reduced; this may even be the cause of misfires of caps, primers, and detonators. Its sensitivity is increased with the size of the crystals. High storage temperatures cause it to be less sensitive; this likewise may be sufficient to cause a detonator or primer cap to become so insensitive that it will not fire. If unconfined it burns fiercely, and when only slightly confined it detonates. The explosive action is very sudden and violent. Its property of igniting, then rapidly detonating, gives a powerful disrupting effect and makes it useful in initiating detonations. It must not be stored with other explosives, for an explosion of even a relatively small amount of fulminate may cause detonation of a large amount of other explosive in the vicinity. (b) Manufacture.-Fulminate of mercury is made by dissolving a charge of pure mercury in nitric acid and then pouring this solution into alcohol. A violent action results which, when completed, leaves the solid crystalline fulminate in the liquid. The charge is then screened, washed, bagged, and placed under water for storage. (c) Uses.-Fulminate of mercury is used in fulminate caps, usually mixed with potassium chlorate, antimony sulphide, and glass, to ignite powder charges, either directly, as in small arms, or through black powder ignition charges contained in the same primer with the cap. It is also used in detonators of all kinds to initiate the detonation of high explosives either directly or through boosters. To give greater safety in handling the amount of fulminate in detonators is generally reduced and replaced with TNT, tetryl, or trinitrobenzene; |
 |
 |
 |
| UNDER CONSTRUCTION THERE IS A LARGE DRAWING THAT IS SORT OF A PICTURE OF THIS PROCESS THAT I WILL AT A LATER DATE SCAN WITH ONE OF MY LARGE SCANNERS AND THEN POST HERE. |